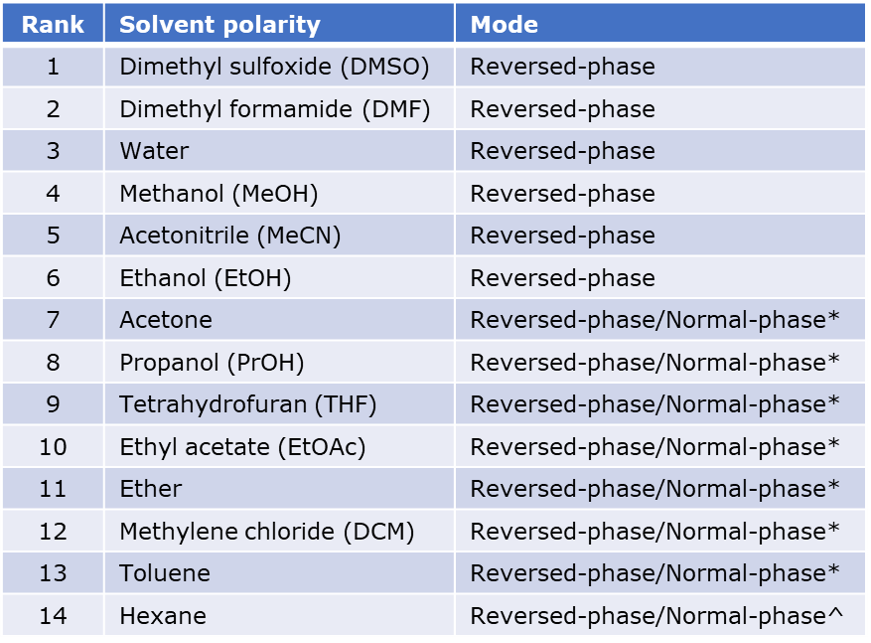
Pharmacognosy for all - Polarity index table of commonly used solvents To calculate the polarity index of a mixture of solvents : Polarity index of the mix.= (Polarity index of solvent 1 *
![PDF] Absorption of Carbon Dioxide into Polar Solvents of 2-Amino-2-Methyl-1- Propanol | Semantic Scholar PDF] Absorption of Carbon Dioxide into Polar Solvents of 2-Amino-2-Methyl-1- Propanol | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/678ef636c67b1127de337d0354c89fcba57cb87f/4-Table3-1.png)
PDF] Absorption of Carbon Dioxide into Polar Solvents of 2-Amino-2-Methyl-1- Propanol | Semantic Scholar
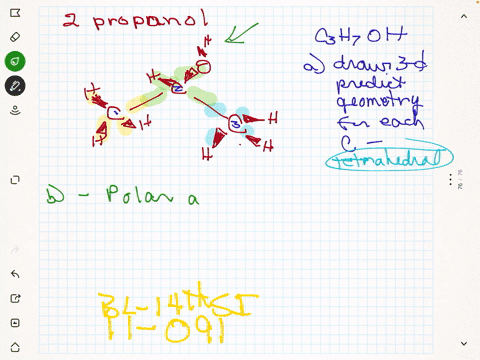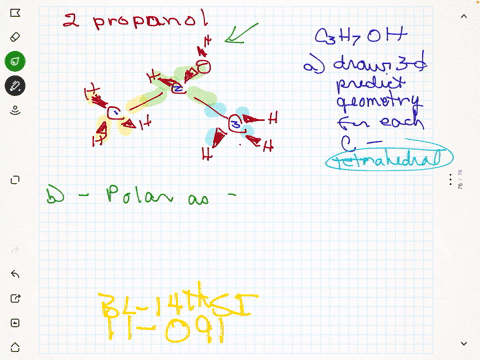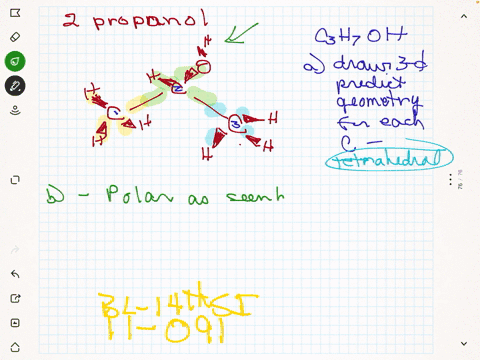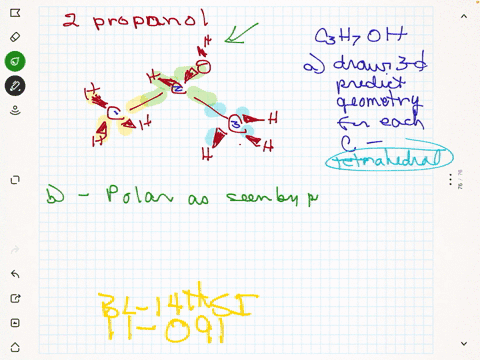
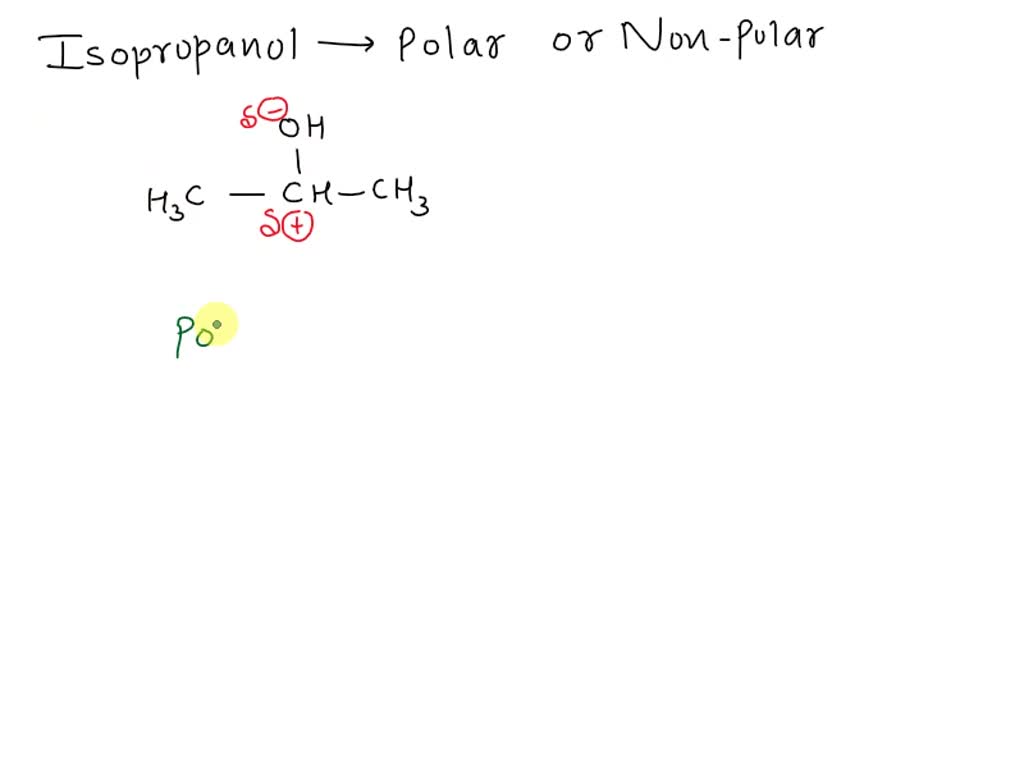
SOLVED:Rubbing alcohol is the common name for 2 -propanol (isopropanol), C3 H7 OH. (a) Draw the three-dimensional structure of the 2 -propanol molecule and predict the geometry around each carbon atom. (b)

Is 1-propanol an ionic, molecular nonpolar, or molecular polar compound? What intermolecular forces are present? | Homework.Study.com

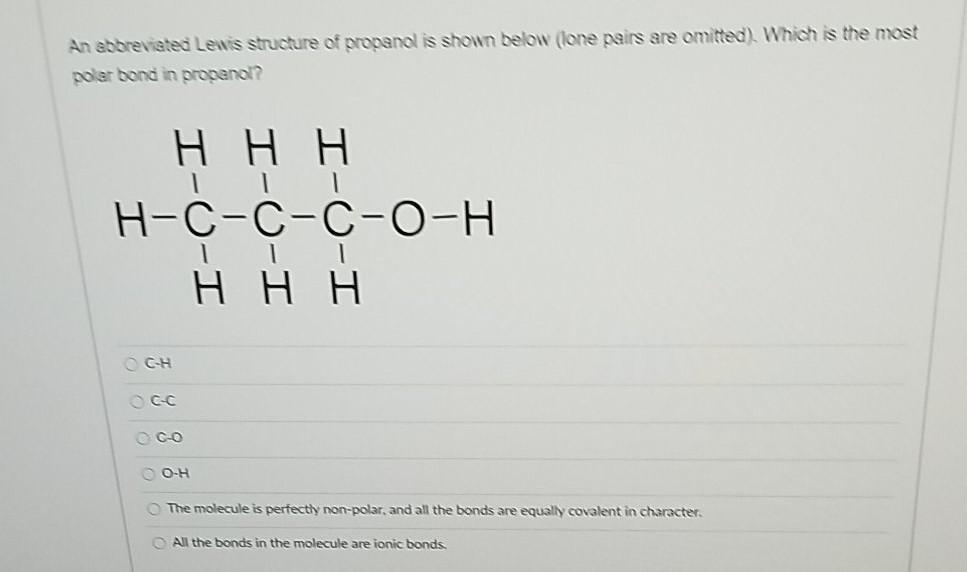
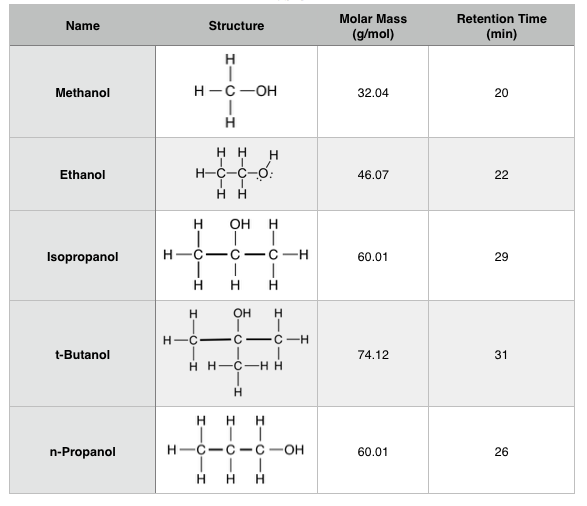
Which of these would have the greatest polarity? I know its one of the propanols but which one? : r/chemhelp