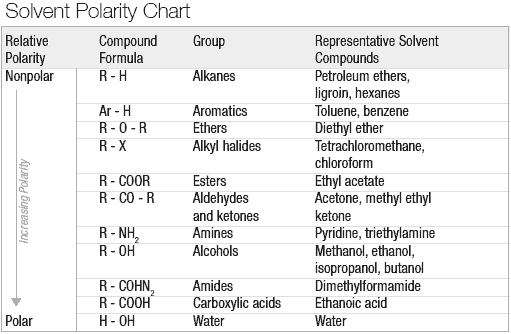
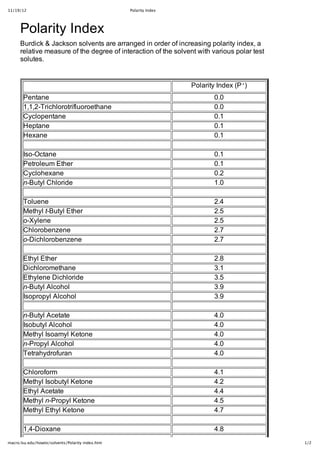
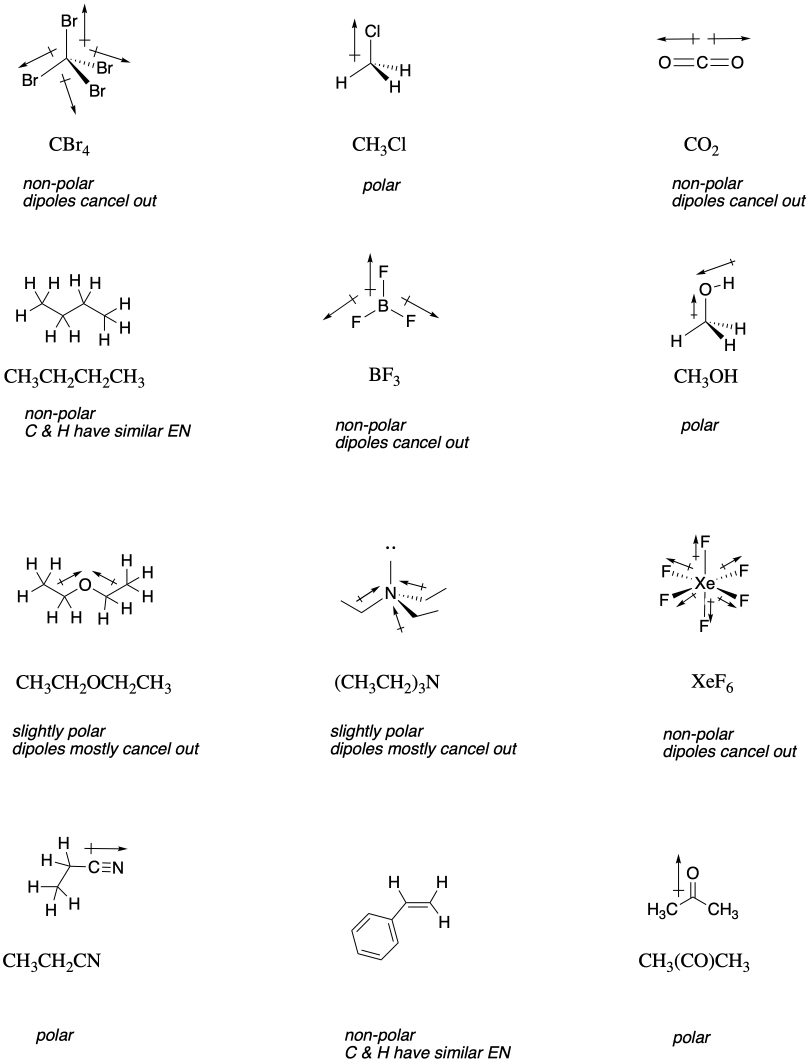
Explain why triethylamine, (CH_3CH_2)_3N, is very slightly soluble in water but readily soluble (with stirring) in 1.5 M HCl. | Homework.Study.com

Switchable-Polarity Solvents Prepared with a Single Liquid Component | The Journal of Organic Chemistry
![SOLVED: Triethylamine [(CH3CH2)3N] bond angle is a molecule in which the nitrogen atom is hybridized and the C-N-C bond angle is 109.5 degrees. Which of the following molecules are polar? CH3OCH3 CF4 SOLVED: Triethylamine [(CH3CH2)3N] bond angle is a molecule in which the nitrogen atom is hybridized and the C-N-C bond angle is 109.5 degrees. Which of the following molecules are polar? CH3OCH3 CF4](https://cdn.numerade.com/ask_images/7f1f04db73ac41c8b0ba27957002fc2a.jpg)
SOLVED: Triethylamine [(CH3CH2)3N] bond angle is a molecule in which the nitrogen atom is hybridized and the C-N-C bond angle is 109.5 degrees. Which of the following molecules are polar? CH3OCH3 CF4

Table 3 from Classification of organic solvents revisited by using the COSMO-RS approach. | Semantic Scholar
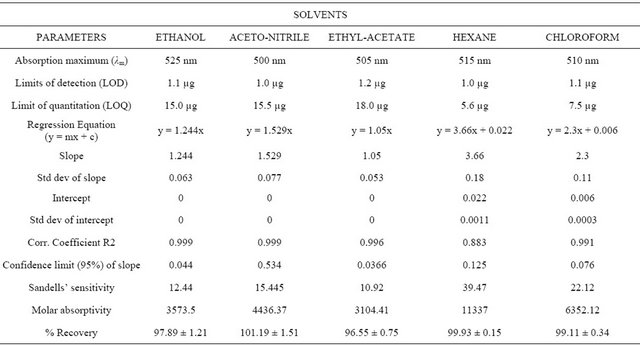
Influence of Solvent Polarity on the Physio-Chemical Properties and Quantitative Determinations of Tenofovir Disoproxil and Emtricitabine with Chloranilic Acid as Complexing Agent