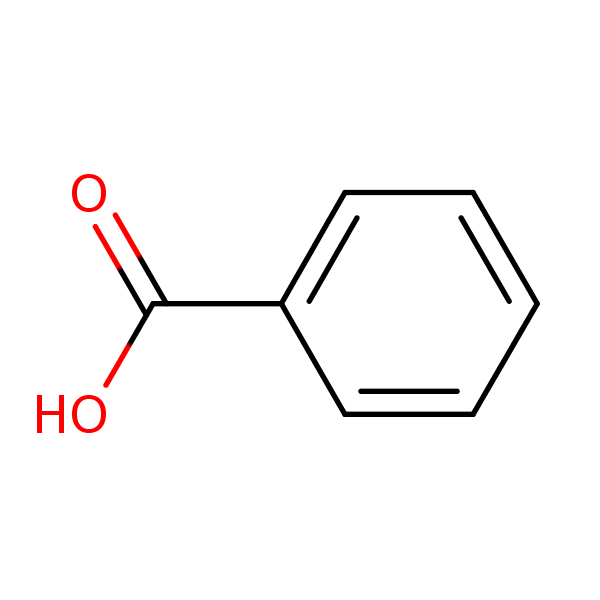
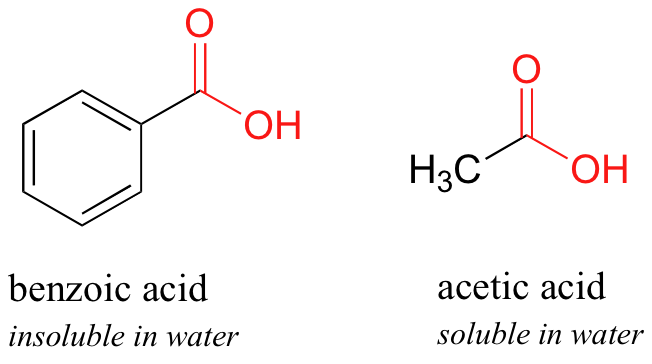
How was a mixture of organic molecules (phenol, benzoic acid, p-toluidine and anisole) separated and isolating process? - Quora
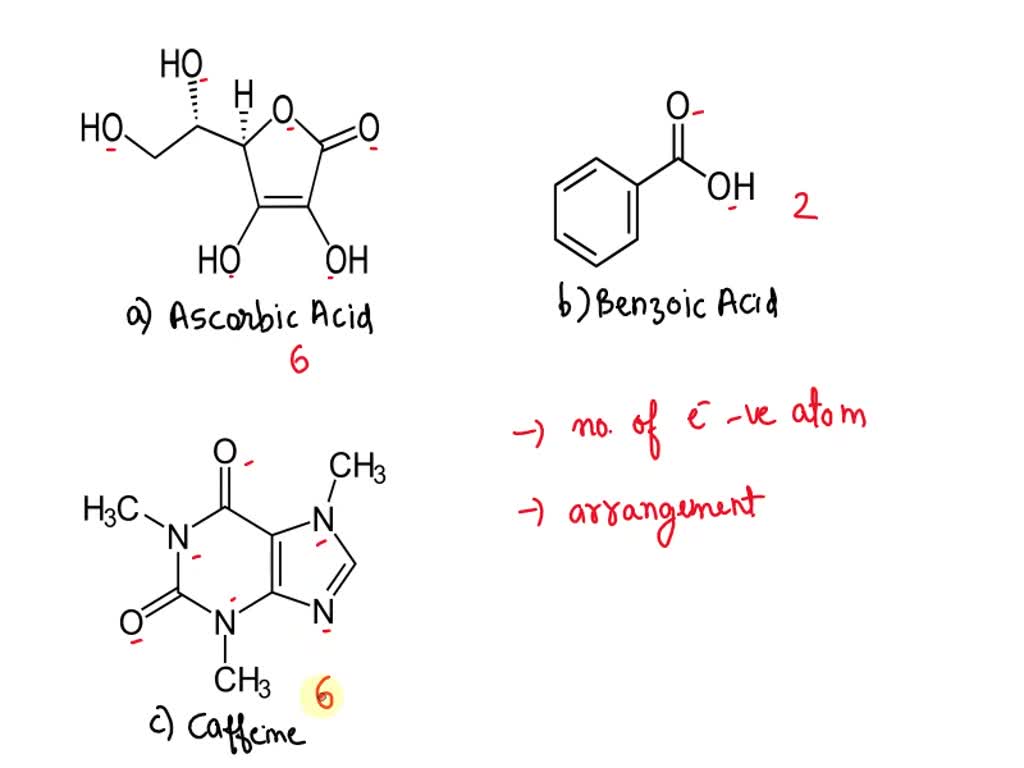
SOLVED: Please rank the following molecules from most polar to least polar. a) ascorbic acid b) benzoic acid c) caffeine

Measured 20 log P values for benzoic acid and aniline derivatives of... | Download Scientific Diagram

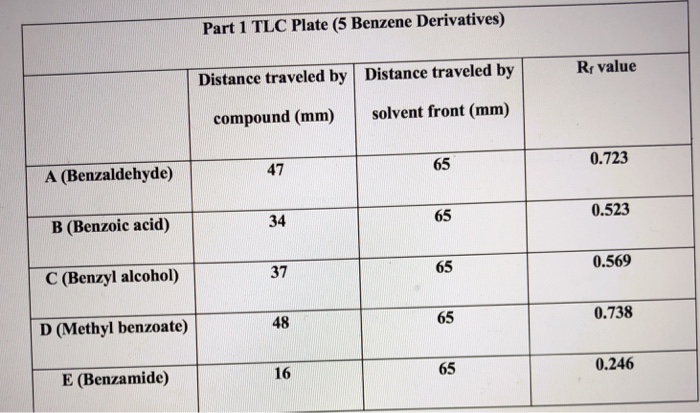
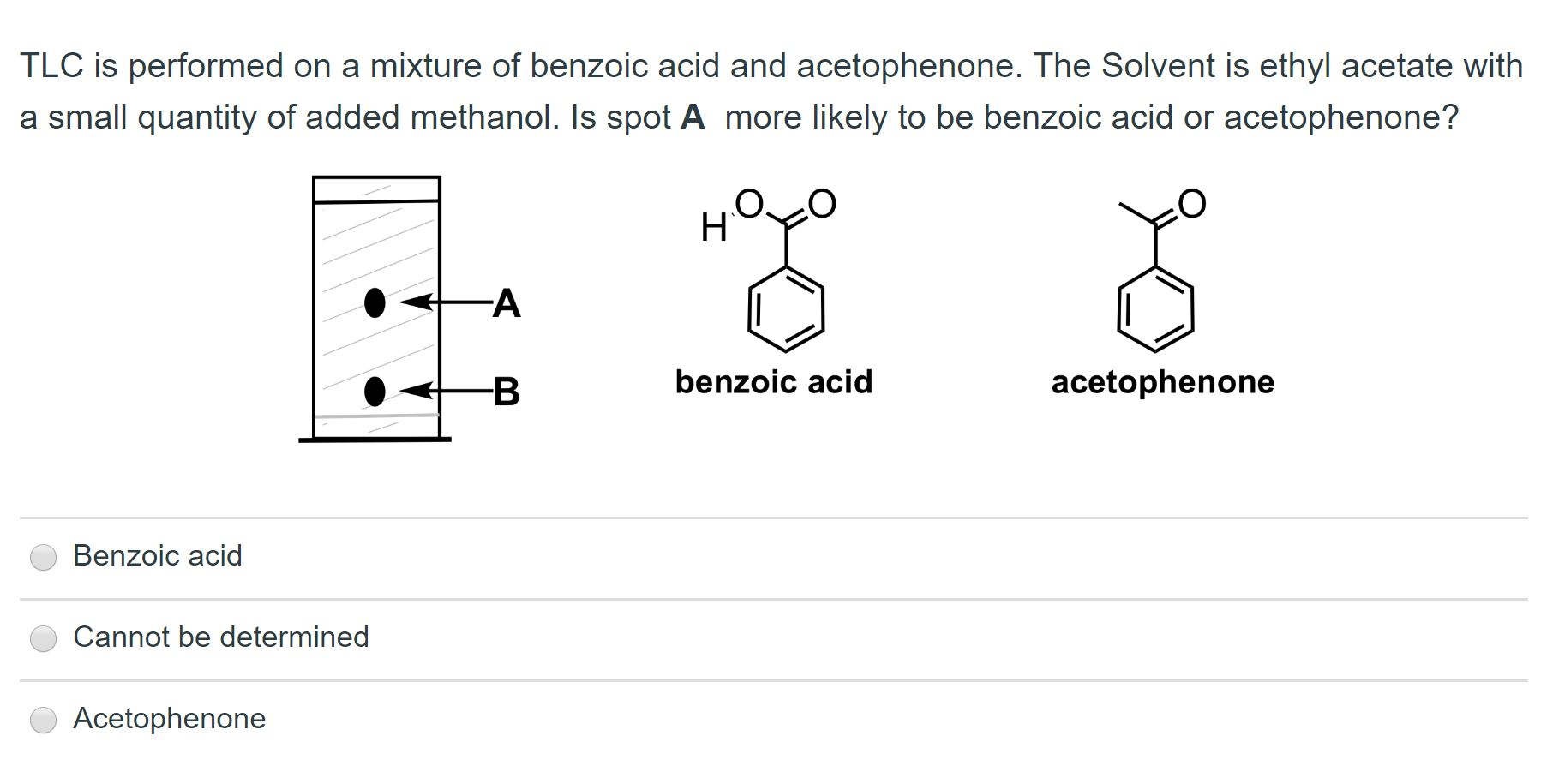
OneClass: The sample is a mixture of three compounds: benzoic acid, benzophenone, and 3-nitrobenzoic ...

2. Draw the chemical structure of benzoic acid and use it to answer the following solubility-based questions: a) Circle and label the region of the molecule that exhibits non-polar behavior. b) Thes

1) Rank trans-stilbene, benzoic acid, and fluorenone by polarity (least polar to most polar). Provide an explanation for your proposed ranking. (2) Hexane, ethyl acetate and acetone will be used as
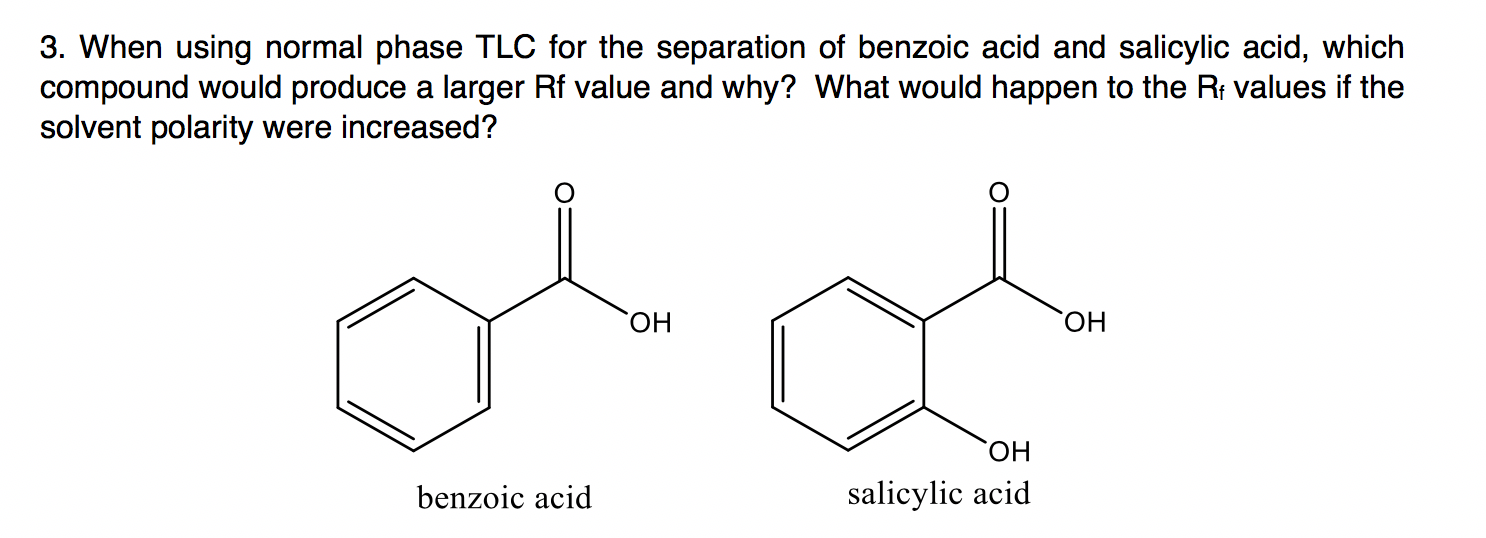
Between acetylsalicylic acid and benzoic acid, which is most polar if you look only at the structure (not at solubility in water g/l), and why? Why is that structure most polar? -