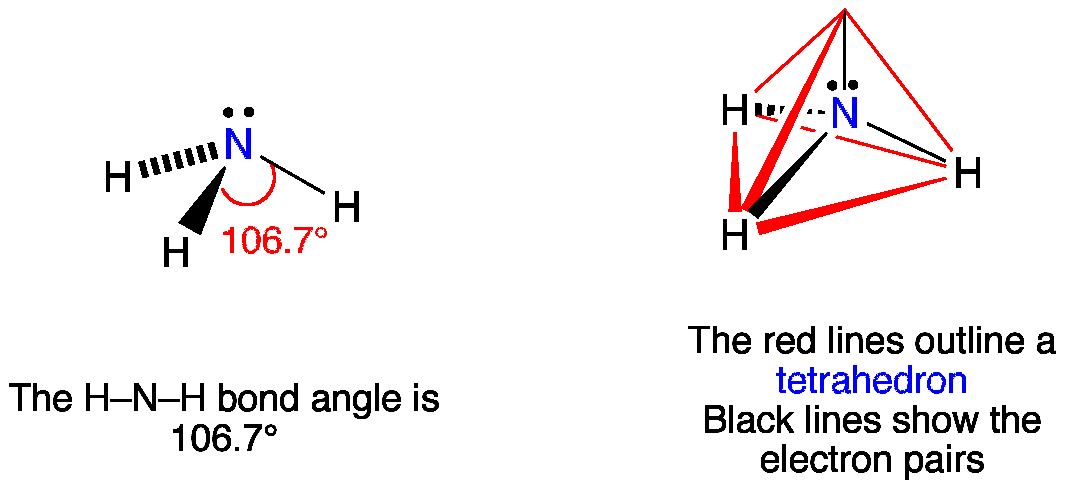
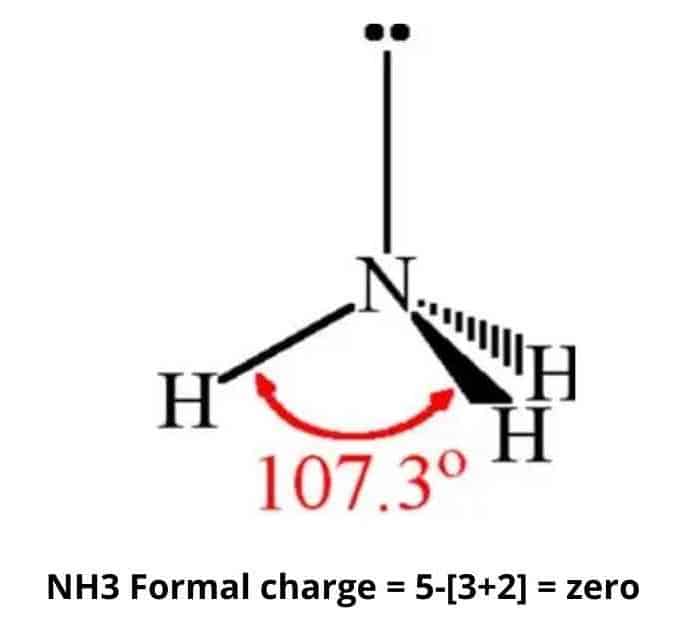
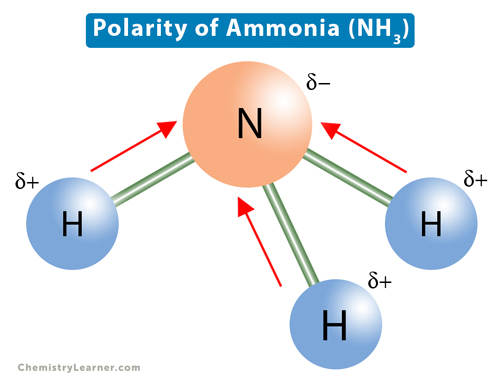
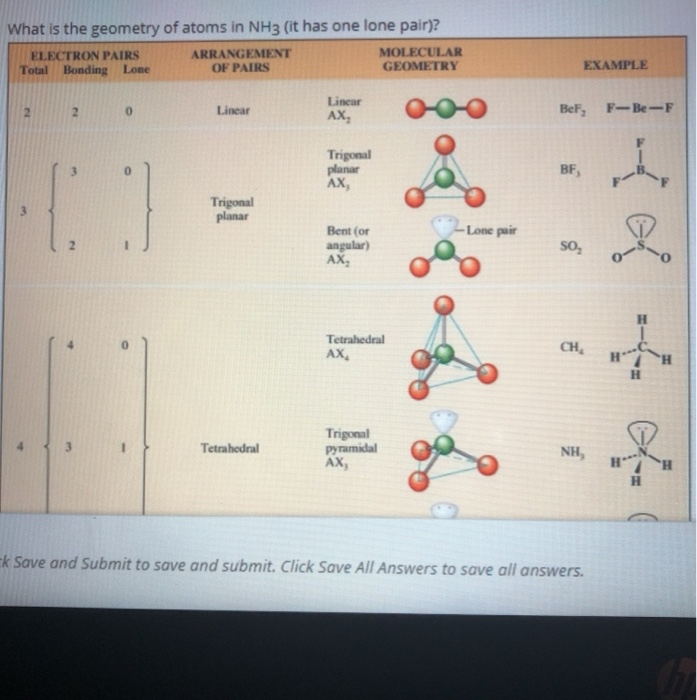
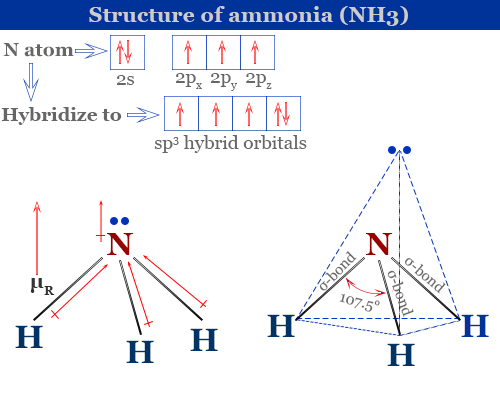
Although the geometries of NH3 and H2O molecules are distorted, the tetrahedral bond angles in water is less than that of ammonia. Why? - Quora

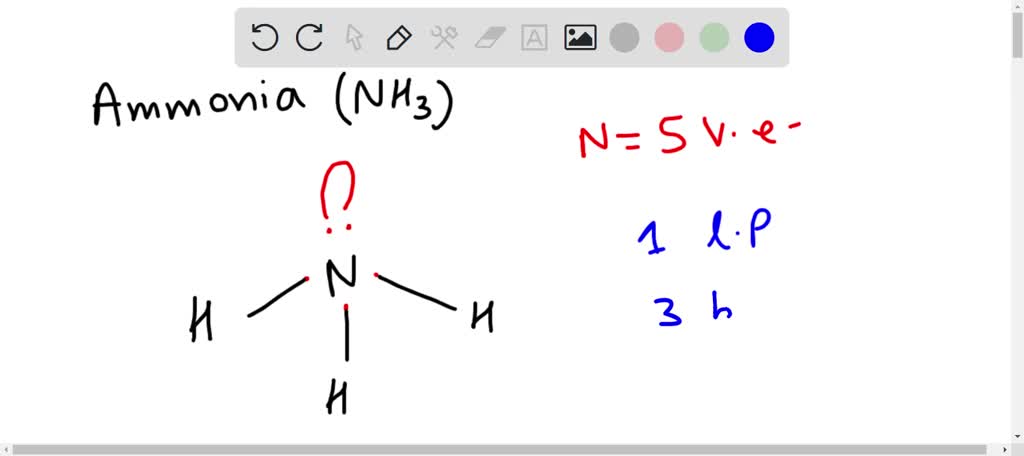
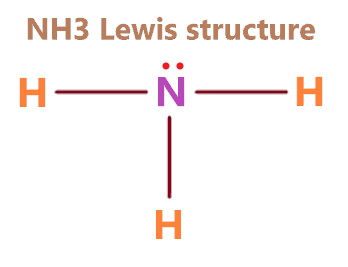
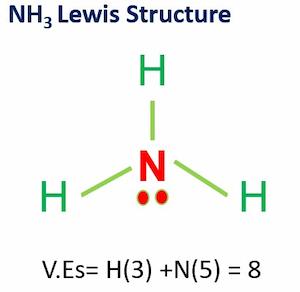
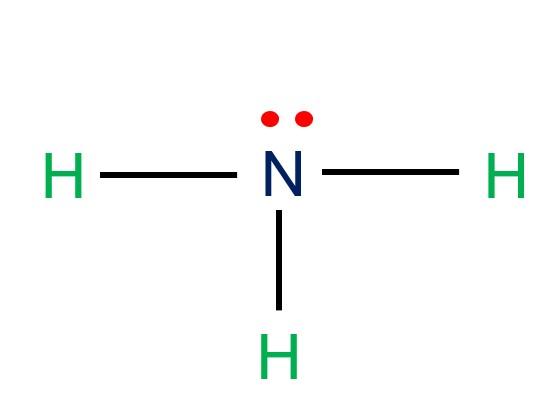
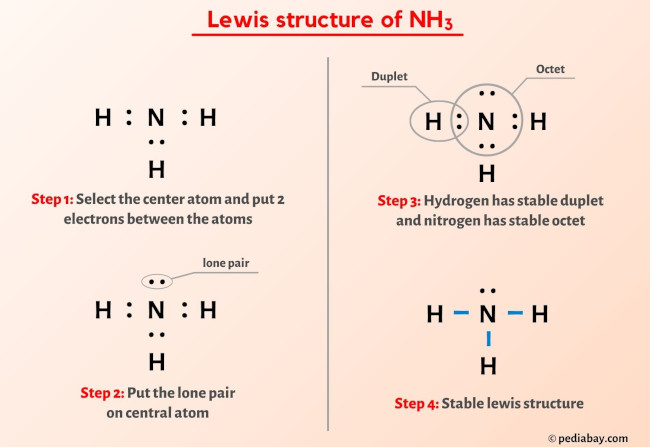
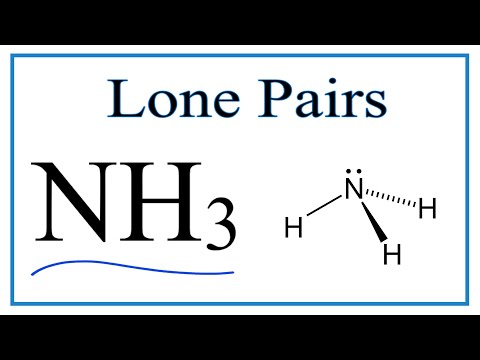
When correctly drawn, the Lewis dot structure for NH3 should have a lone pair of electrons on the central N. True False | Homework.Study.com

Why bond angle in water is less that that of ammonia although their geometries are distorted tetrahedral?

SOLVED: Draw the Lewis electron dot diagram for ammonia (NH3) on a sheet of scrap paper. You may consult the periodic table on Zoom to draw your diagram. Which of the following