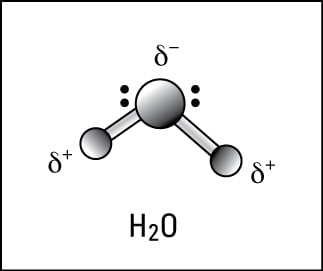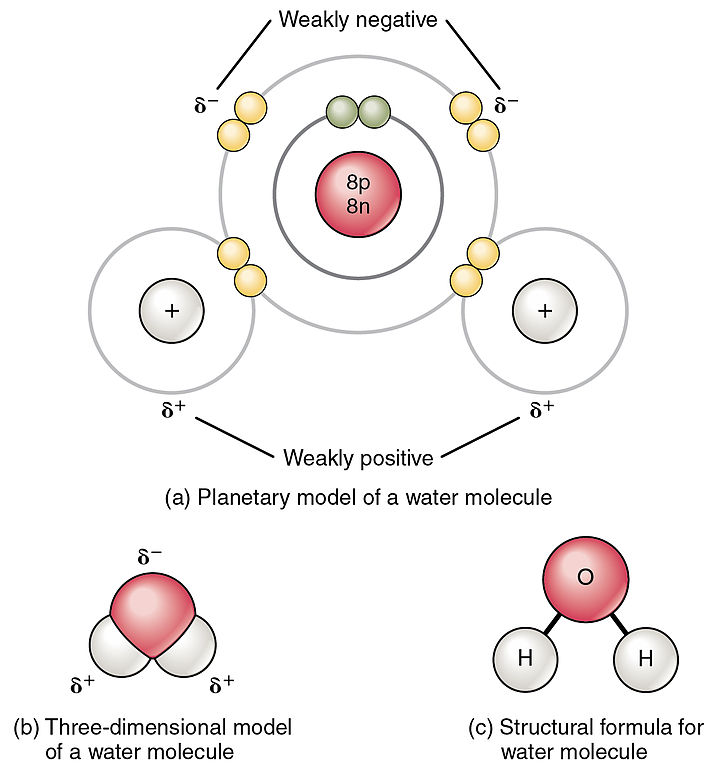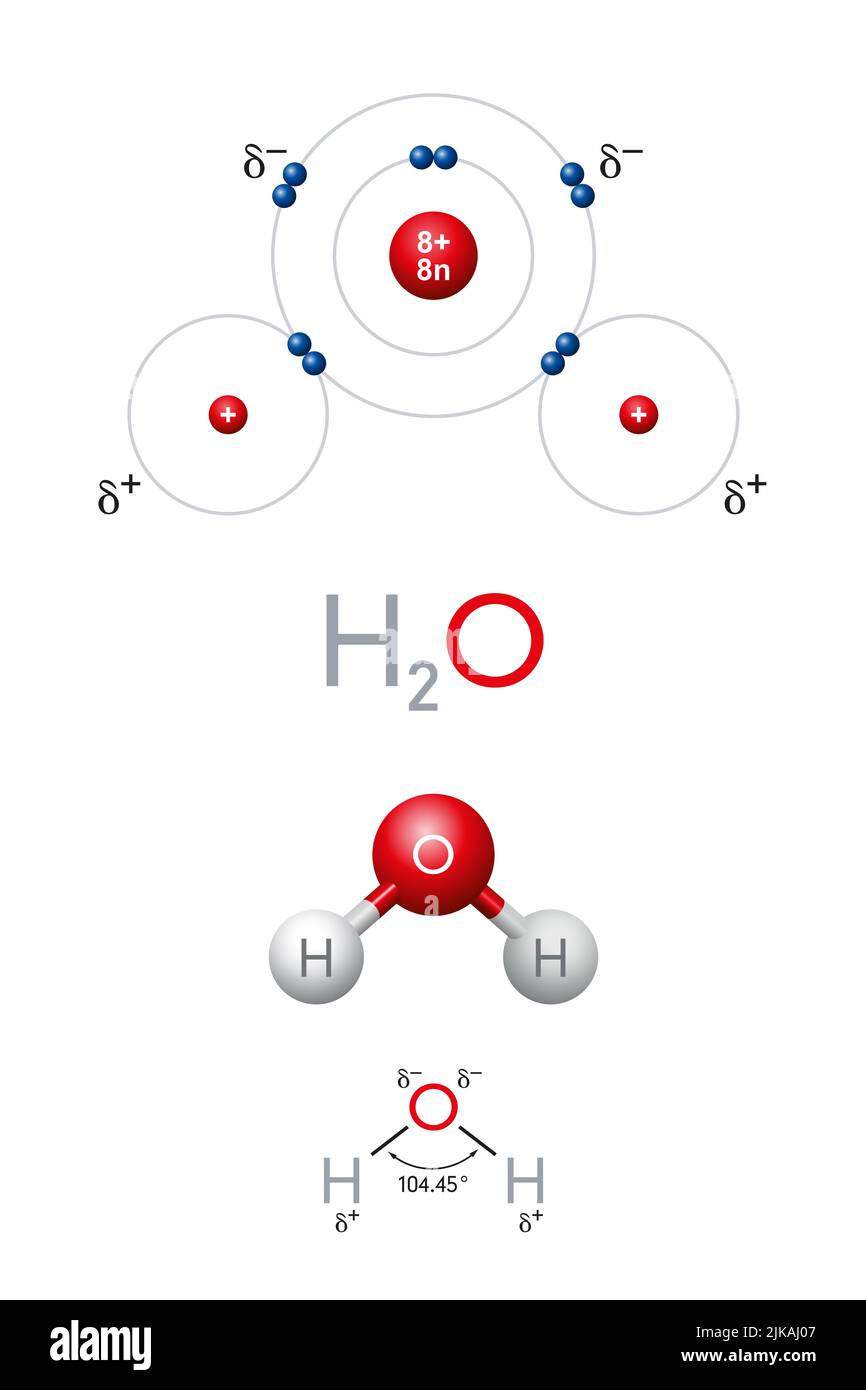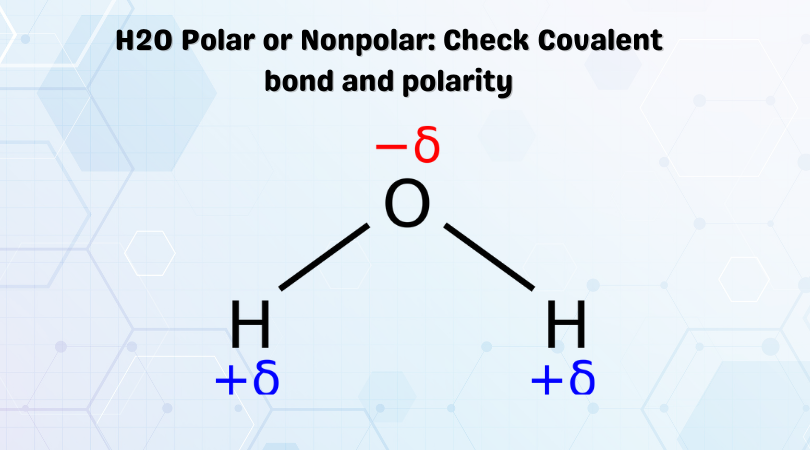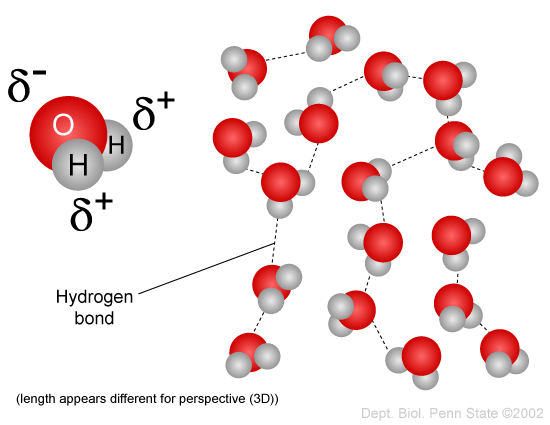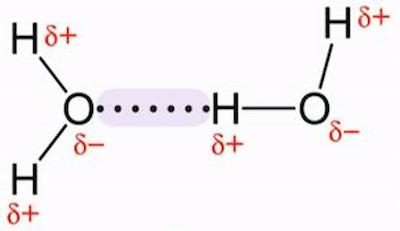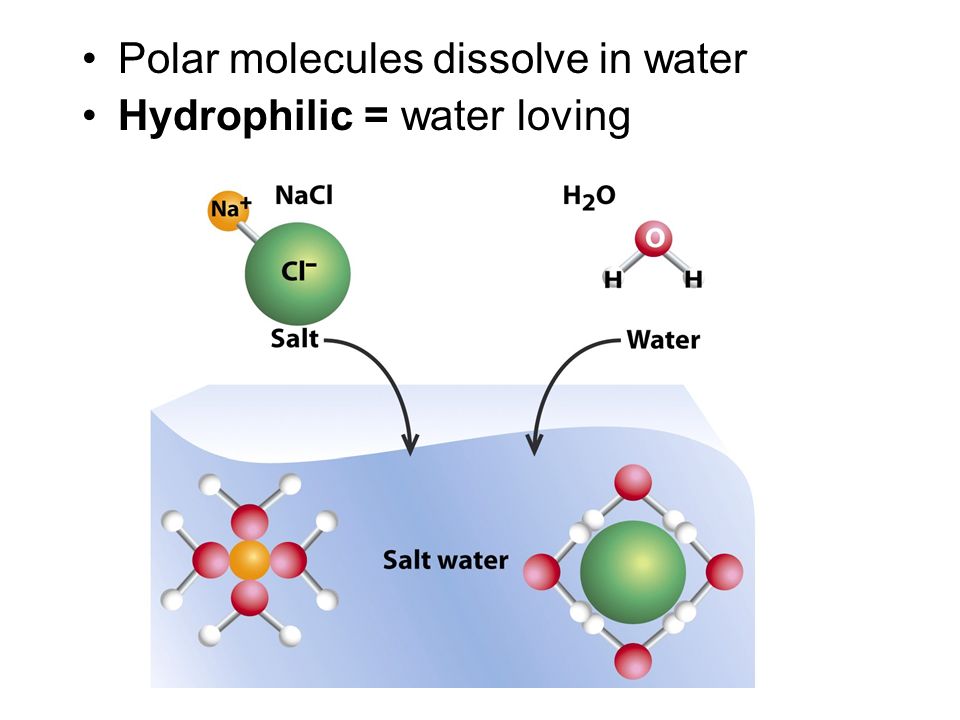
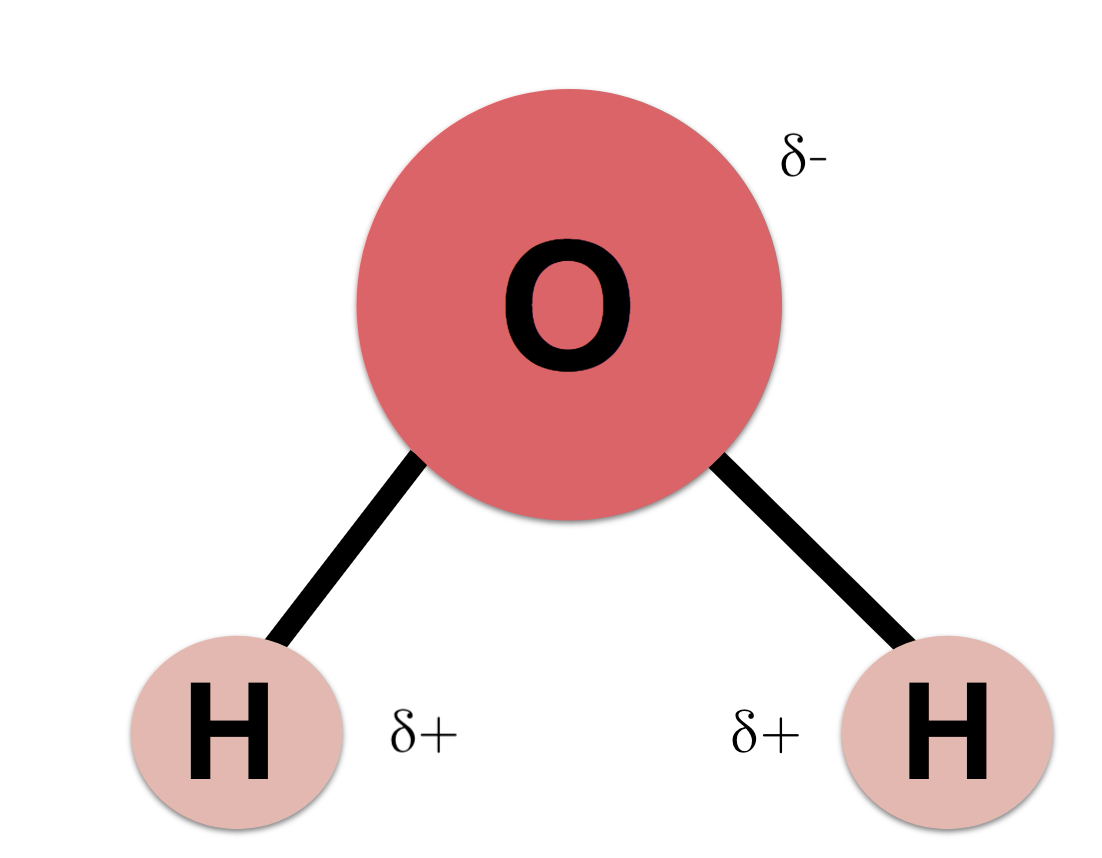
O H - H - Water (H 2 O) Water is a polar molecule This means that... A.All atoms have equal electrical charges B.A water molecule is linear, - ppt download

Water (H2O) molecules are bent. Based on your knowledge of shape and polarity, how would you expect two water molecules to interact? Is there anything in your experience with or knowledge of



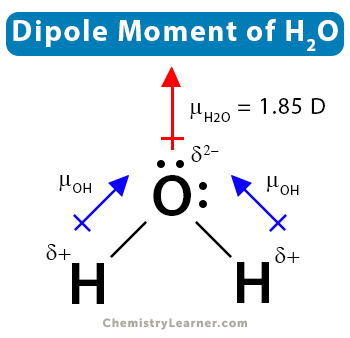

:max_bytes(150000):strip_icc()/PolarConvalentBond-58a715be3df78c345b77b57d.jpg)