
Are All Polar Molecules Hydrophilic? Hydration Numbers of Ketones and Esters in Aqueous Solution | The Journal of Physical Chemistry B

Polarity‐Reversed Allylations of Aldehydes, Ketones, and Imines Enabled by Hantzsch Ester in Photoredox Catalysis - Qi - 2016 - Angewandte Chemie - Wiley Online Library

A chemoselective polarity-mismatched photocatalytic C(sp3)–C(sp2) cross-coupling enabled by synergistic boron activation | Catalysis | ChemRxiv | Cambridge Open Engage

Relative reactivity and polarity of carboxylic acid derivatives. X = halide | Download Scientific Diagram

The optimal ester and ketones candidates in terms of polarity and T b . | Download Scientific Diagram


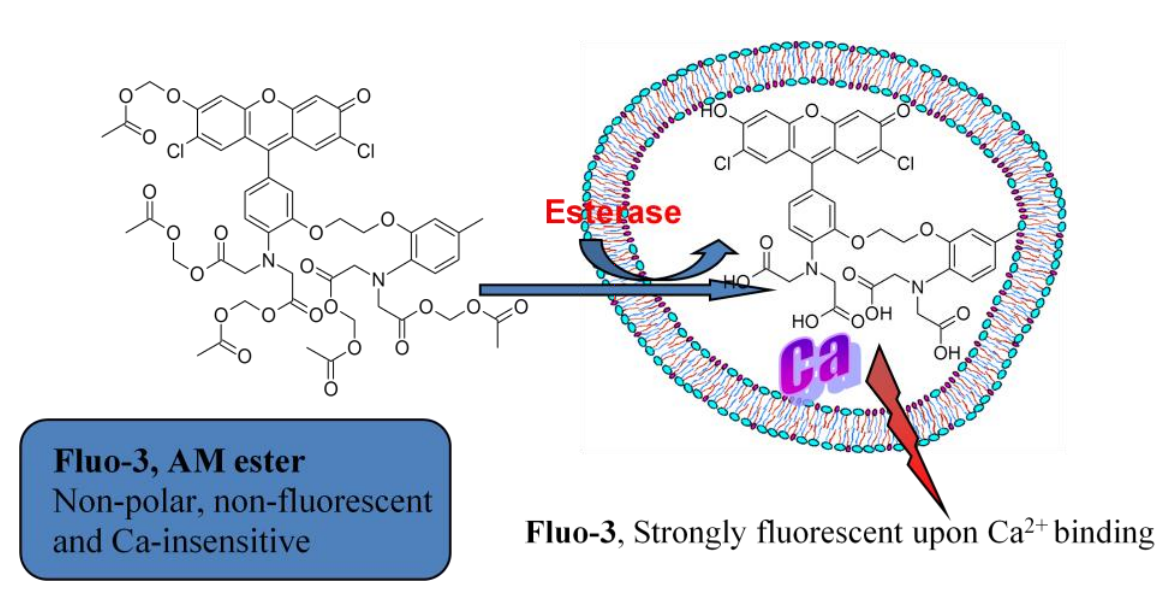
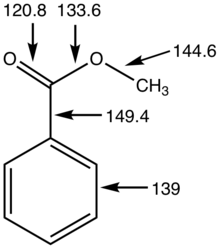

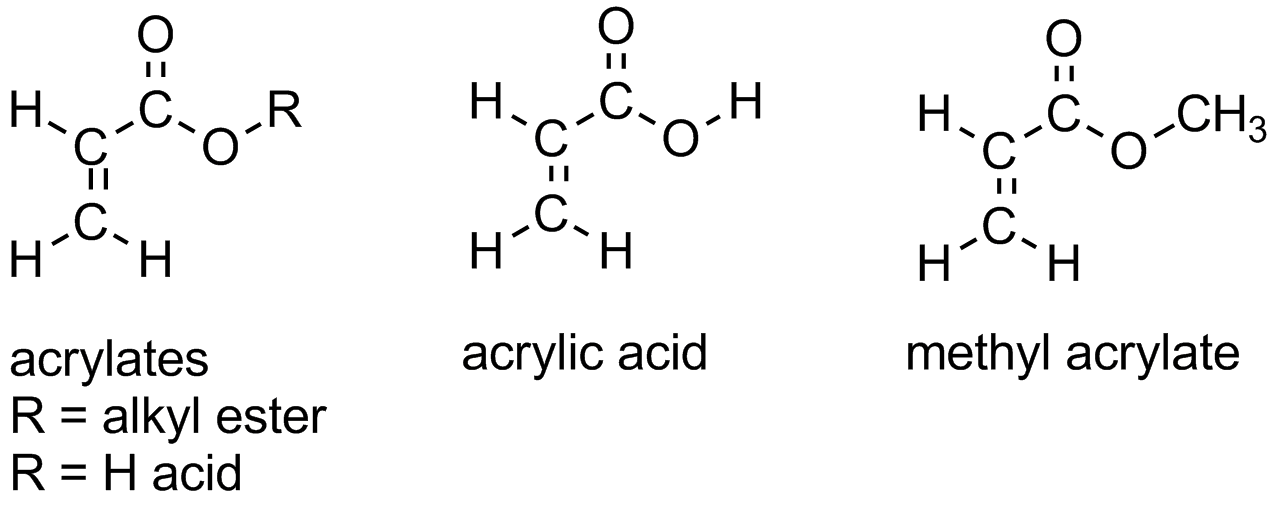
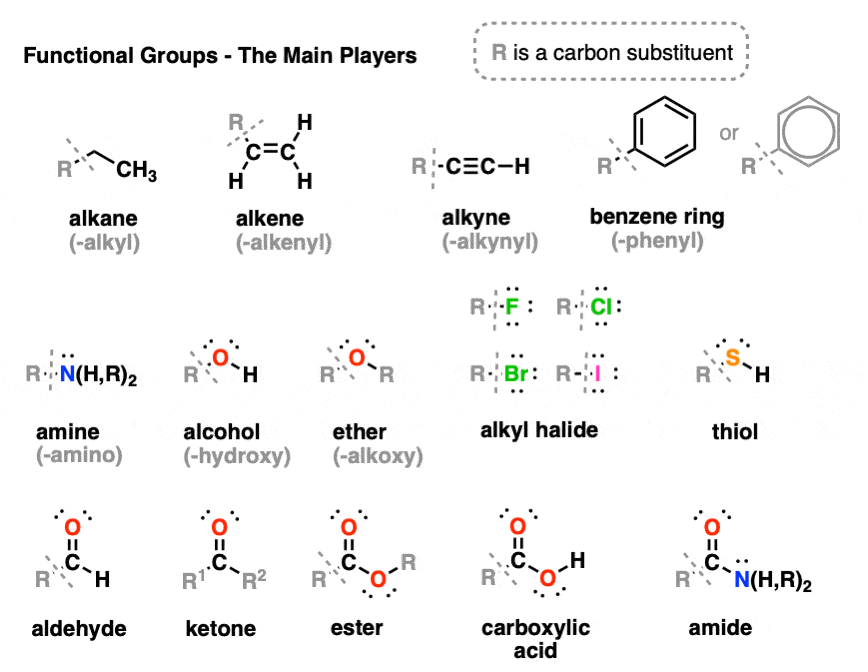


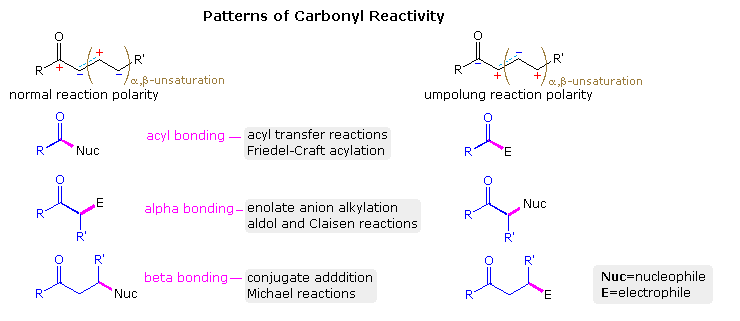


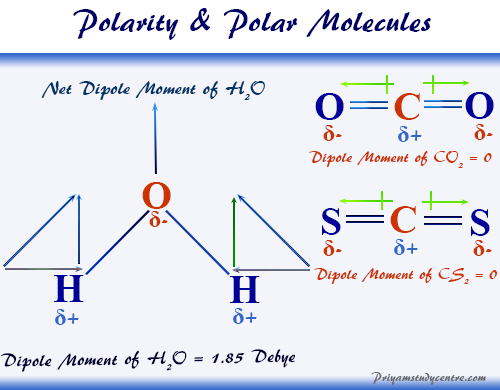

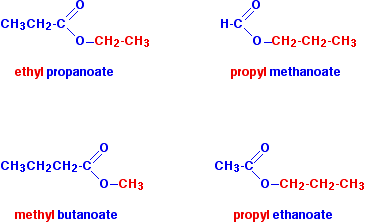
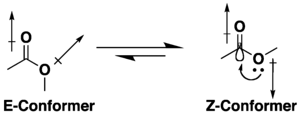

:max_bytes(150000):strip_icc()/ester-59134cd83df78c9283519859.png)

![PDF] Dielectric Study on Polar Binary Mixtures of (Ester-Alcohol) at 298.2 K | Semantic Scholar PDF] Dielectric Study on Polar Binary Mixtures of (Ester-Alcohol) at 298.2 K | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/22d27867e332bbb9a798f544c96974a9cccd9cd4/2-Table1-1.png)