
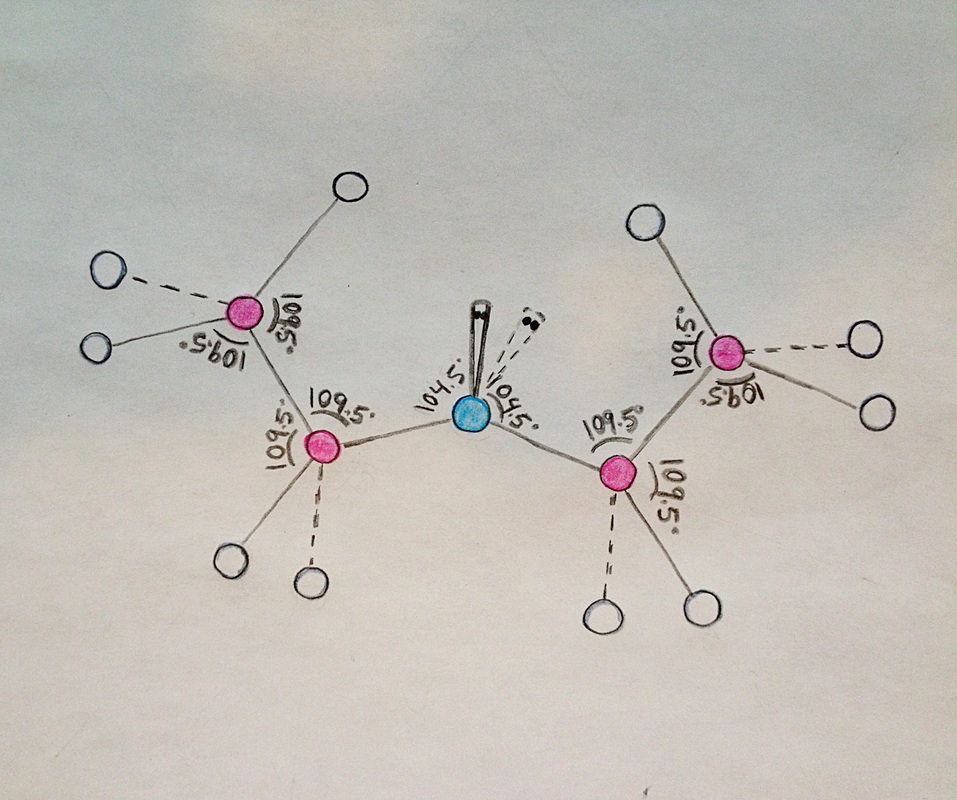
Diethyl ether has a much higher boiling point than butane despite having a higher molecular weight. Explain why this is the case, making reference to the molecular structures of both compounds.

How can benzoic acid dissolve into diethyl ether since benzoic acid is polar and diethyl ether is non-polar? | Homework.Study.com

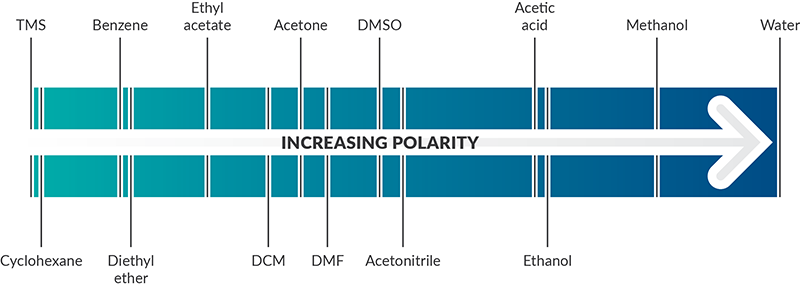
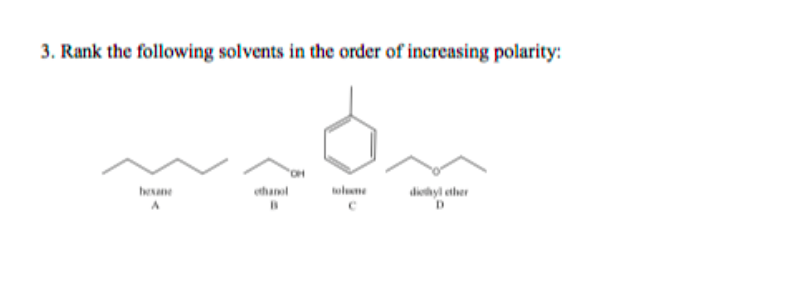
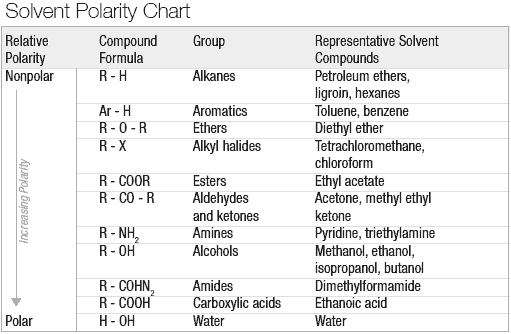
Arrange the following solvents in order of increasing polarity: a) ethanol b) ethyl acetate c) petroleum ether d) toluene e) acetone | Homework.Study.com










![Q.62. Question: Although diethyl ether... [FREE SOLUTION] | Vaia Q.62. Question: Although diethyl ether... [FREE SOLUTION] | Vaia](https://s3.eu-central-1.amazonaws.com/studysmarter-mediafiles/media/textbook-exercise-images/image_lV0jd6p.png?X-Amz-Algorithm=AWS4-HMAC-SHA256&X-Amz-Credential=AKIA4OLDUDE42UZHAIET%2F20230813%2Feu-central-1%2Fs3%2Faws4_request&X-Amz-Date=20230813T115116Z&X-Amz-Expires=90000&X-Amz-SignedHeaders=host&X-Amz-Signature=a415badc663c0d8753fbcefe33befba4a3fc2e295623baf5bbe1e60ba049078a)


