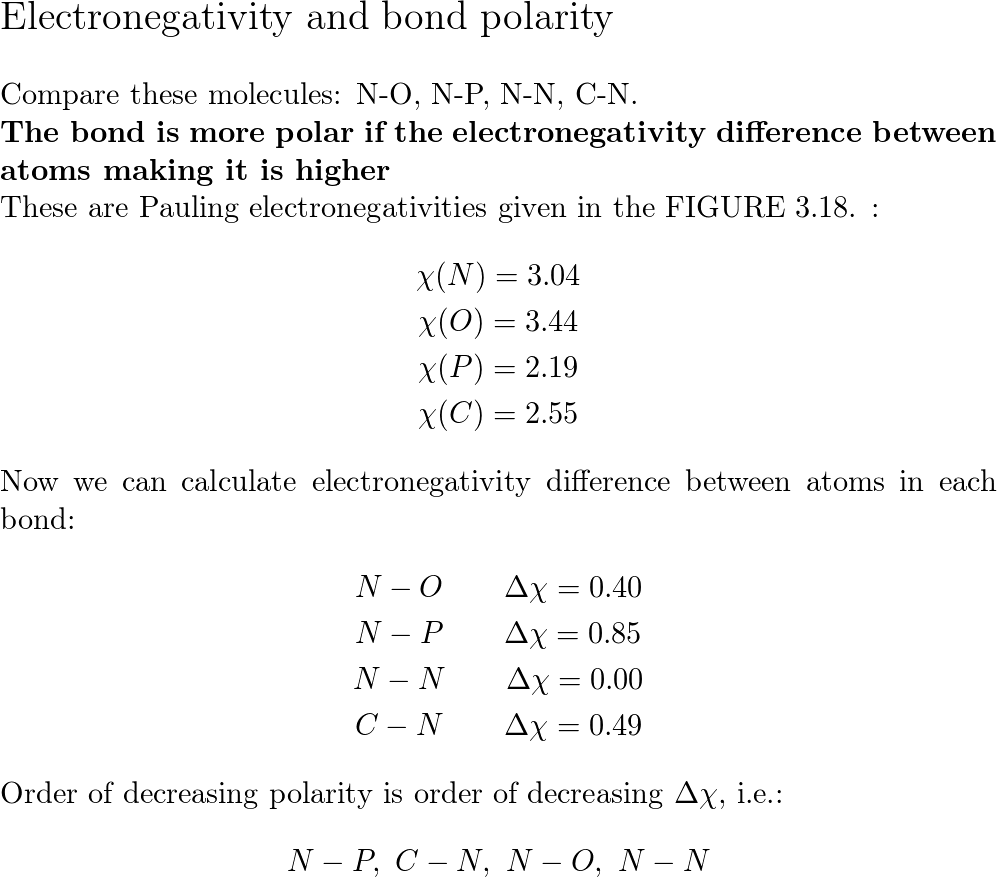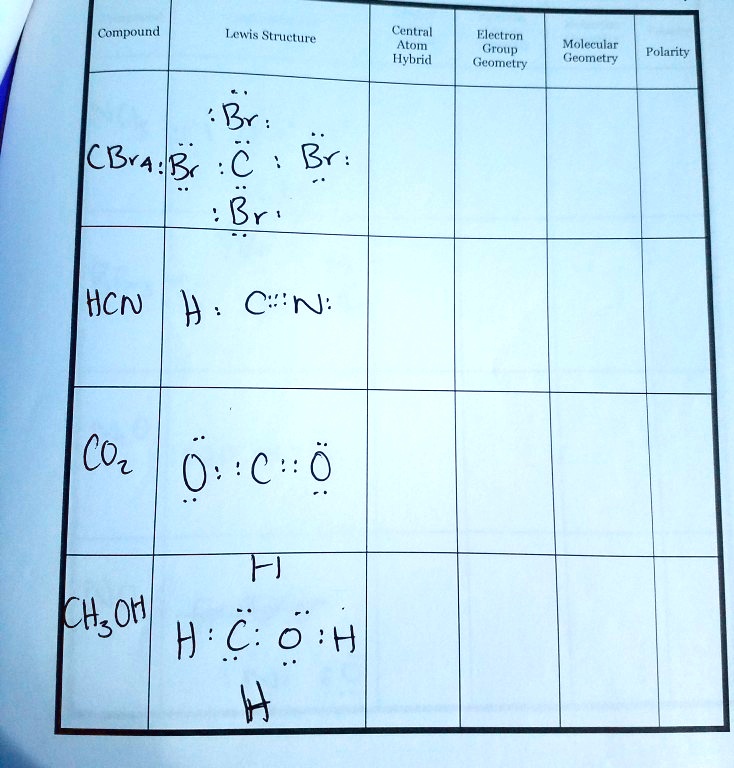
SOLVED: Compound Lewis Structure Central Atom Hybridization Molecular Geometry Polarity Br: HCN H CN: CO2 KHOX H2O CBr4
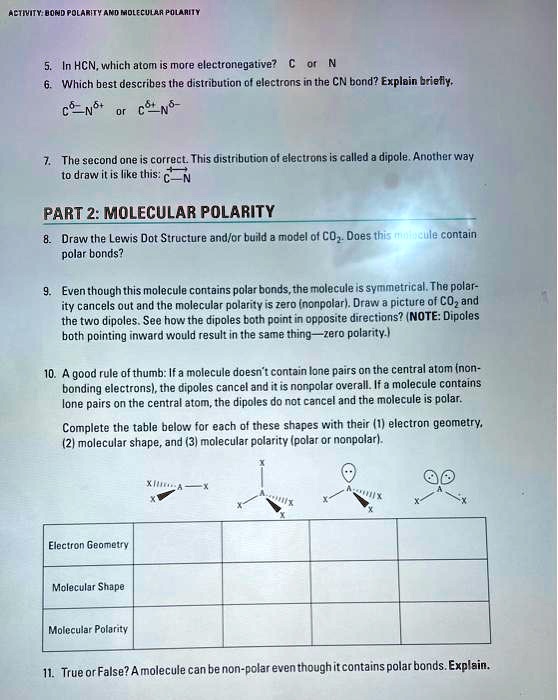
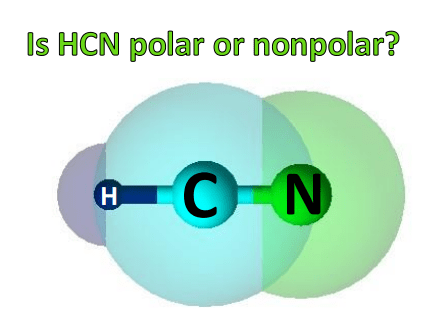
SOLVED: Trityl-methanol Molecular POLARITY In HCN, which atom is more electronegative? Which best describes the distribution of electrons in the CN bond? Explain briefly. The second one is correct. This distribution of

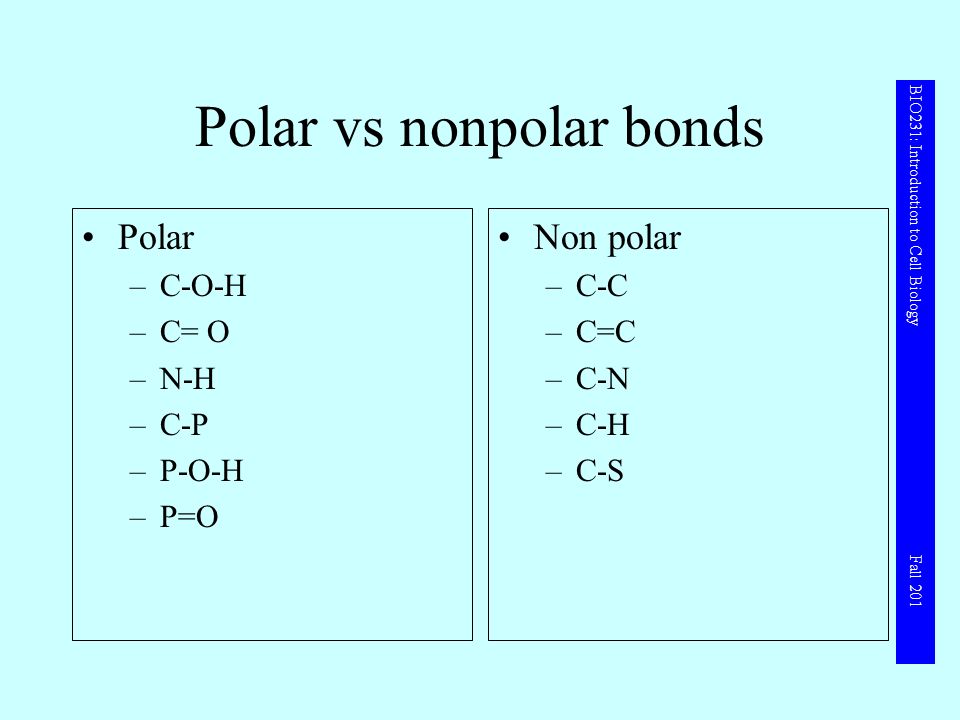
SOLVED: Write Lewis structures and predict whether each of the following is polar or nonpolar: HOCN (exists as HO-CN), COS, XeF2, CF2Cl2, SeF6, H2CO (C is the central atom).

The cyanide ion `CN and N_(2)` are isoelectronic, but in contrast to `CN^(-), N_(2)` is chemically - YouTube

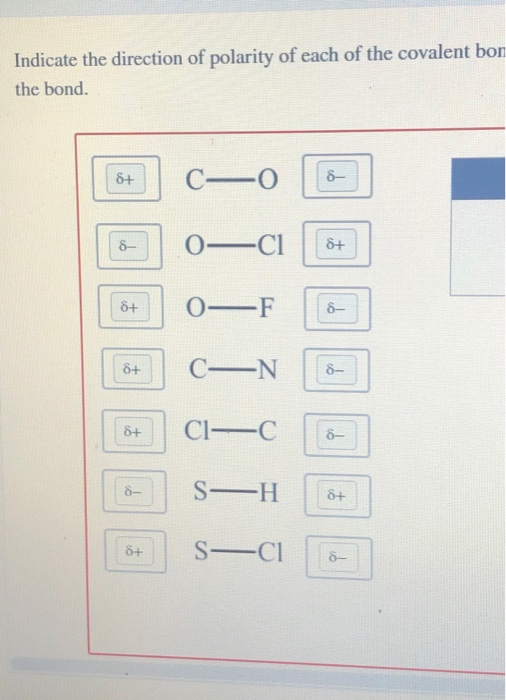
Indicate the direction of polarity of each of the covalent bonds by placing the appropriate delta notation - Brainly.com

Cyanide Ion (CN-): Lewis Structure, Molecular Geometry, Hybridization, MOT And Polarity | Scientific Sarkar

The cyanide ion, CN^ - and N2 are isoelectronic, but in contrast to CN^ - , N2 is chemically inert, because of :