
Explain why the dipole moment of chlorobenzene is lower than cyclohexyl chloride ? – The Unconditional Guru

Effect of polarity and temperature on the binary interaction between D2EHPA extractant and organic solvents (kerosene, n-heptane, chlorobenzene and 1-octanol): Experimental and thermodynamics - ScienceDirect
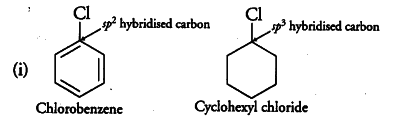
Explain why the dipole moment of chlorobenzene is lower than that of cyclohexyl chloride? - CBSE Class 12 Chemistry - Learn CBSE Forum
Which of the following molecule is polar:A.\n \n \n \n \n B.\n \n \n \n \n C.\n \n \n \n \n D.All of these

The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride. CBSE 2010C / 2000 - YouTube

Table 1 from Self-Assembly of a Strong Polyhedral Oligomeric Silsesquioxane Core-Based Aspartate Derivative Dendrimer Supramolecular Gelator in Different Polarity Solvents. | Semantic Scholar
Explain why: (a) The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride? - Sarthaks eConnect | Largest Online Education Community

Figure 2 from A dispersive liquid-liquid microextraction using a switchable polarity dispersive solvent. Automated HPLC-FLD determination of ofloxacin in chicken meat. | Semantic Scholar








![Kannada] Why the dipole moment of chlorobenzene is lower than that of Kannada] Why the dipole moment of chlorobenzene is lower than that of](https://d10lpgp6xz60nq.cloudfront.net/physics_images/OSW_SP_CHE_XII_C10_E01_018_S01.png)

![Telugu] Explain why the dipole moment of chlorobenzene is lower than Telugu] Explain why the dipole moment of chlorobenzene is lower than](https://d10lpgp6xz60nq.cloudfront.net/physics_images/VIK_CHE_QB_C11_E02_016_S01.png)


