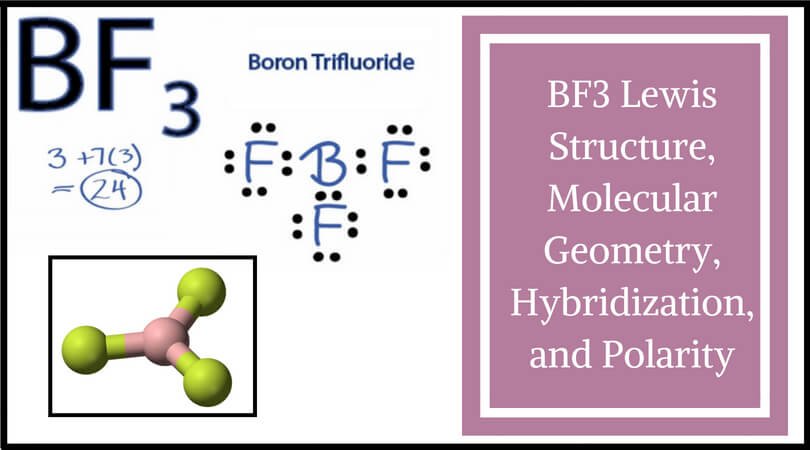
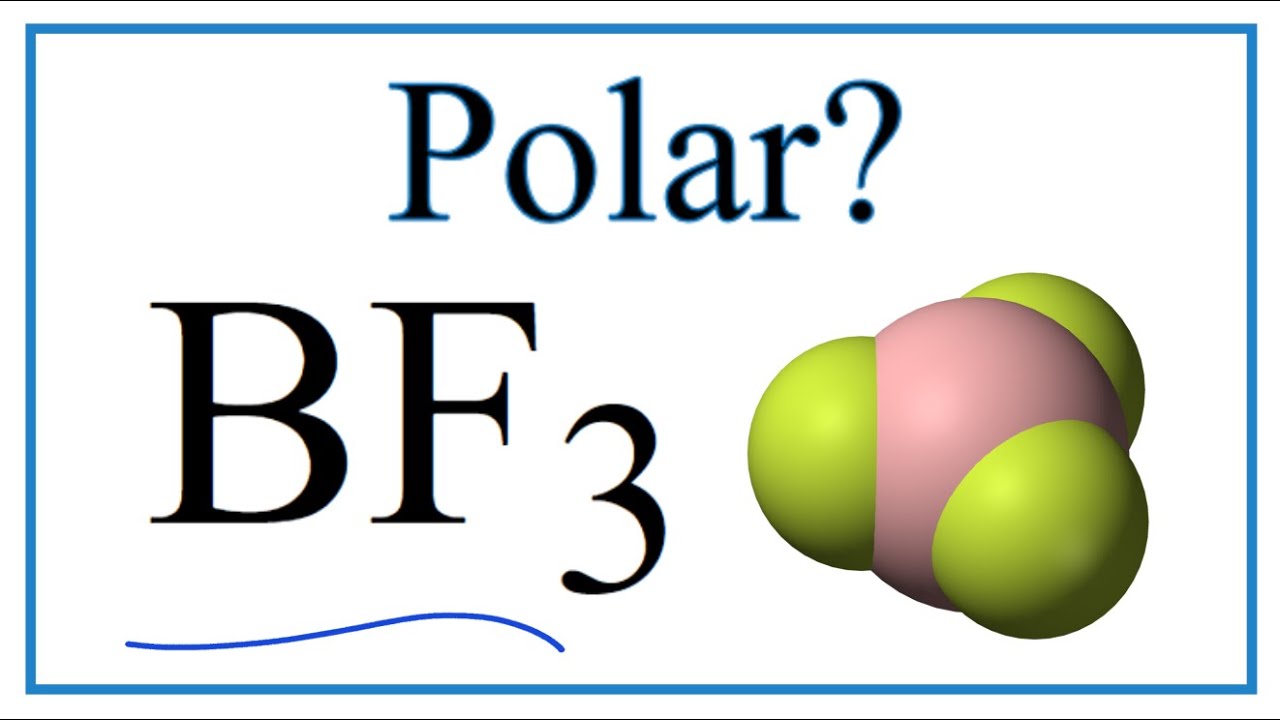
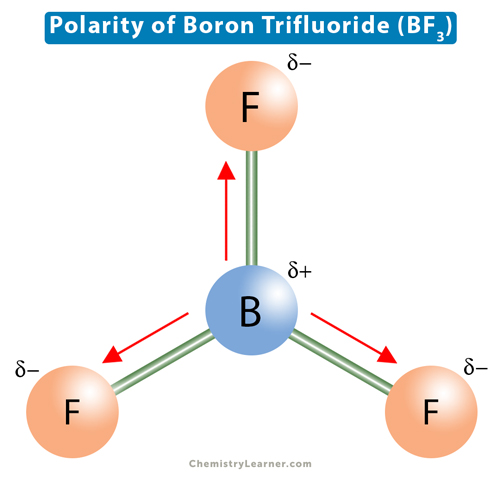
Ammonia (NH3) is a polar molecule while boron trifluoride (BF3), is a nonpolar molecule. What is the difference in the polarity of these compounds? - Quora

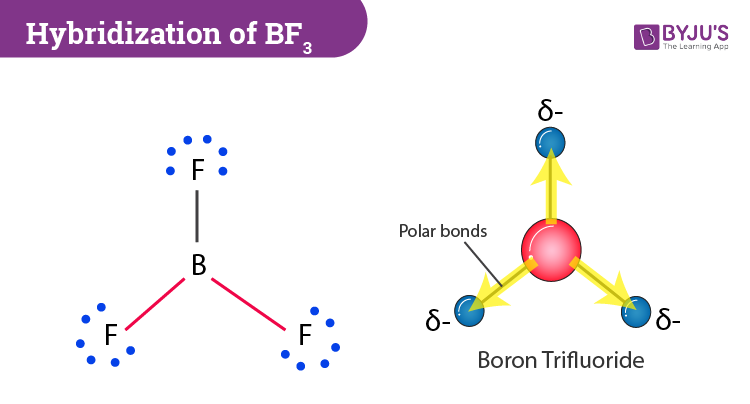
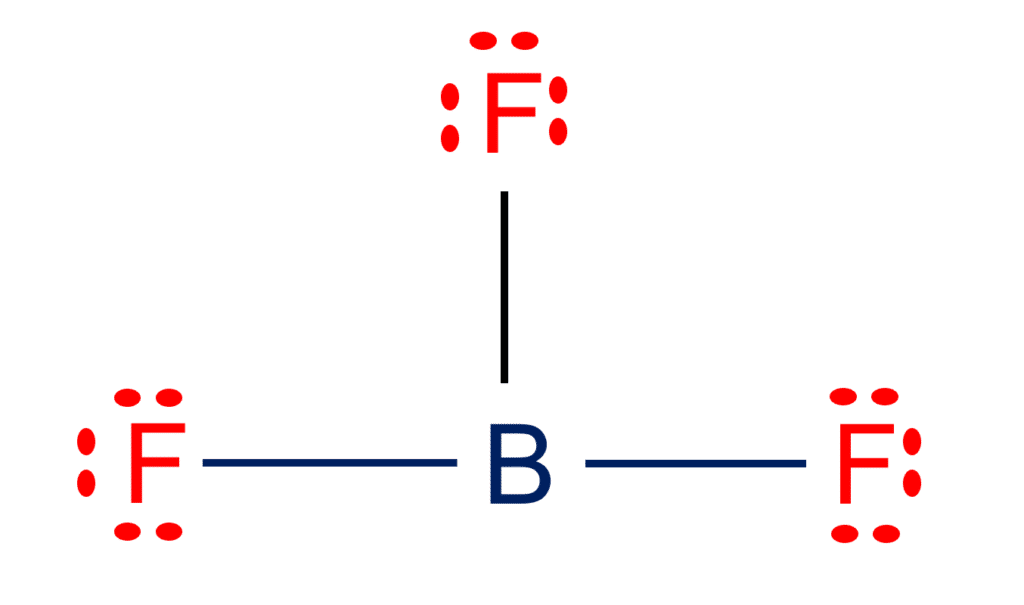
BF3 and NF3 both are covalent compounds but NF3 is polar whereas BF3 is non - polar. This is because :
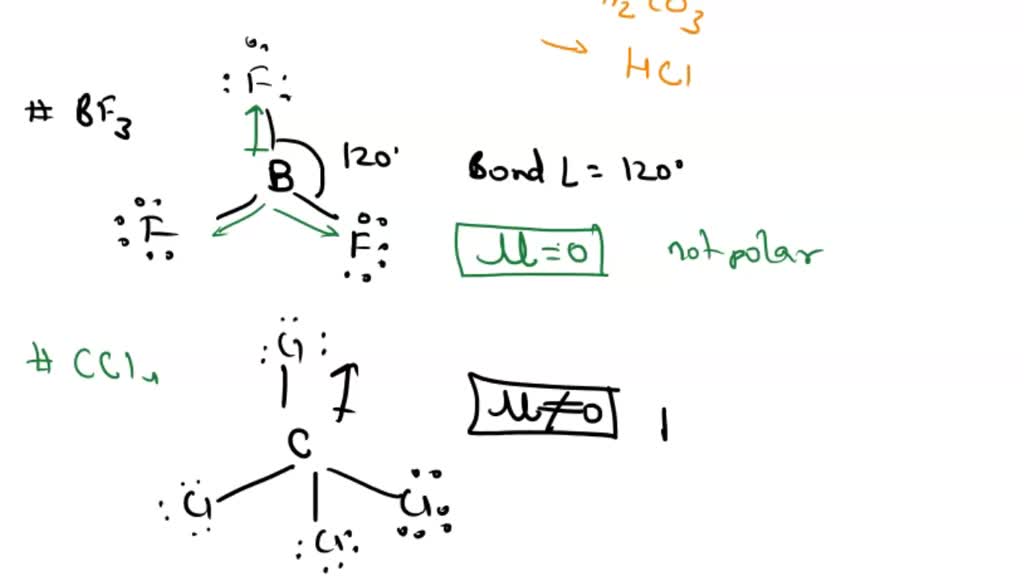
SOLVED: Determine the bond polarity, molecular polarity; electron geometry molecular geometry and bond angles and Lewis structure: 1 BF3 2 CCI 3 PCI; HCO 5 HCI